Non-Compartmental Analysis (and publication list referencing PCModfit, lower down this page)
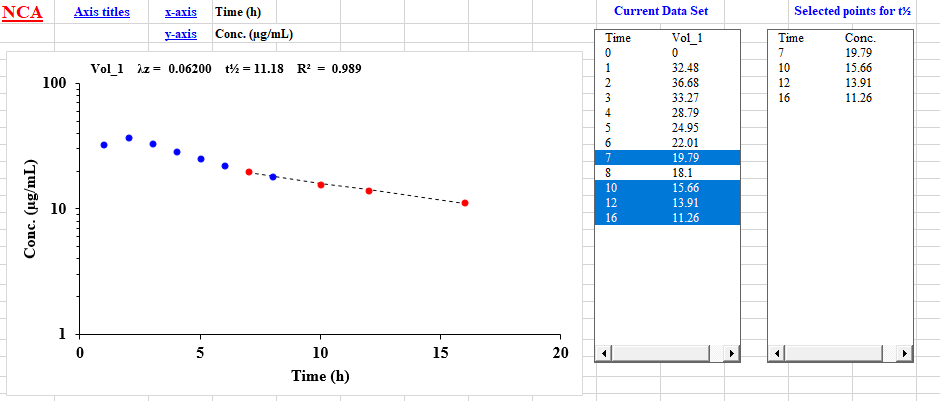
The model independent option (NCA) in PCModfit allows the user to analyse time-concentration data within Excel. The user can easily select (or deselect) the number of data points interactively using a high resolution graphics window to estimate half-lives, tmax, Cmax and various types of AUC and AUMC. The results are directly and neatly transferred to the NCA Excel spreadsheet and as separate Excel files (for record purposes) so further editing can be conducted. High quality plots of the data and fitted line can easily be imported into Word etc. Both pictures and text results are stored in the 'Results' folder in the PCModfit directory
There are also extensive help facilities within each option. A clipped snippet of what the user is likely to see is shown below for information.The NCA intercept value is now (V7.1 onwards) displayed on the graph, in the Spreadsheet and Excel results files. Useful for C0 values with bolus i.v. data and in other calculations. From V7.1 onwards, additional parameters such as CL, MRT, Vd and Vss are also calculated with all results displayed in an Excel spreadsheet which is saved in directory /Results/ as NCA*.xls or .xlsx files, which opens automatically after the data set(s) are analysed, and in the NCA spreadsheet.
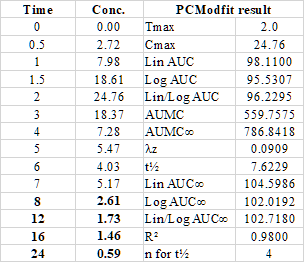
Since the latest version was released (V6 onwards), PCModfit NCA has been tested using published data from the literature and verified
by several users and the all results show the same values as two other programs i.e. WinNonLin and PKSolver.
Ref.
Computer methods and programs in biomedicine 9 9 (2010) 306-314
PKSolver: An add-in program for pharmacokinetic and
pharmacodynamic data analysis in Microsoft Excel
Yong Zhanga,1, Meirong Huoa,1, Jianping Zhoua,*, Shaofei Xieb.
The data and results are shown in the Ref. and the user should try the NCA data for comparison themselves.
Shown below for information...
A quick search of the internet suggests that PCModfit has been referenced numerous times in publications and used in submissions
and without a detailed look, over 100 were found and ca. 70 of these are shown below for information.
1. Enhancing the Bioavailability of the Ellagitannin, Geraniin: Formulation, Characterization, and in vivo Evaluation.
Sumita Elendran, V. Shiva Kumar, Usha Sundralingam, Wai-Kit Tow, Uma Devi Palanisamy.
Elsevier, International Journal of Pharmaceutics. Volume 660, 20 July 2024, 124333
2. Pharmacokinetics of recombinant human annexin A5 (SY-005) in patients with severe COVID-19
Tschirhart BJ, Lu X, Mokale Kognou AL, Martin CM, Slessarev M, Fraser DD, Leligdowicz A, Urquhart B and Feng Q (2024)
ORIGINAL RESEARCH article
Front. Pharmacol., 10 January 2024, 14:1299613.
3. Celecoxib-Loaded Self-micellizing Solid Dispersion Suppresses Its Delayed Absorption in Rats with Impaired Gastrointestinal Motility
Mizuki Ogino, Keisuke Yakushiji, Hiroki Suzuki, Yukinori Yamauchi, Yoshiki Seto, Hideyuki Sato, Satomi Onoue
Chemical and Pharmaceutical Bulletin. 2023 Volume 71, Issue 10 Pages 787-791.
4. Characterization and Pharmacokinetic Assessment of a New Berberine Formulation with Enhanced Absorption In Vitro and in Human Volunteers
November 2023, Pharmaceutics 15(11)
Solnier, Julia, Zhang Yiming, Kuo, Yun, Du, Min, Roh, Kyle, Gahler, Roland, Wood, Simon, Chang, Chuck.
5. Pharmacokinetics and effect on renal function and average daily gain in lambs after castration and tail docking, of firocoxib and meloxicam
K Kongara, G Purchas, VSR Dukkipati, D Venkatachalam, N Ward, H Hunt & D Speed
New Zealand Veterinary Journal. Research article accepted author version posted online: 06 Jul 2023
6. Pharmacokinetics of N,N-dimethyltryptamine in Humans
Meghan Good, Zelah Joel, Tiffanie Benway, Carol Routledge, Chris Timmermann, David Erritzoe, Richard Weaver, Graham Allen, Charlotte Hughes, Helen Topping, Amy Bowman & Ellen James
European Journal of Drug Metabolism and Pharmacokinetics, 48, 311-327 (Issue 3, May 2023).
7. Development of stabilized fuzapladib solution for injection: forced degradation study and pharmacokinetic evaluation
Hideyuki Sato, Chika Yamane, Koji Higuchi, Takeshi Shindo, Hiroshi Shikama, Kohei Yamada, Satomi Onoue.
Pharmaceutical Development and Technology, 09 Jun 2022
8. Neuropsychopharmacological profiling of scoparone in mice
Joanna Kowalczyk, Barbara Budzynska, Lukasz Kurach, Daniele Pellegata, Nesrine S. El Sayed, Jurg Gertsch and Krystyna Skalicka-Wozniak
Nature: Scientific Reports volume 12, Article number: 822 (2022)
9. Pharmacokinetics and milk extraction pattern of eprinomectin at different dose rates in dairy cattle
Mariana Ballent, Candela Canton, Paula Dominguez, Laura Mate, Laura Ceballos, Carlos Lanusse, Adrian Lifschitz
Veterinary Pharmacology and Therapeutics, Sept. 2021, 45, 92-98.
10. Metabolic Soft Spot and Pharmacokinetics: Functionalization of C-3 Position of an Eph-Ephrin Antagonist Featuring a Bile Acid Core as an Effective Strategy to Obtain Oral Bioavailability in Mice
Francesca Ferlenghi, Carmine Giorgio, Matteo Incerti, Lorenzo Guidetti, Paola Chiodelli, Marco Rusnati, Massimiliano Tognolini, Federica Vacondio, Marco Mor, Alessio Lodola
Pharmaceuticals 2022, 15(1), 41
11. Stem-cell mobilization of healthy sibling donors with pegfilgrastim-A prospective open-label phase II trial (EudraCT no: 2005-004971-39)
Vladan Vucinic, Madlen Jentzsch, Sabine Leiblein, Enrica Bach, Yvonne Remane, Kai Schulze-Forster, Michael Cross, Wolfram Ponisch, Sebastian Schwind, Georg-Nikolaus Franke, Uwe Platzbecker, Dietger Niederwieser
Transfusion. Dec. 2021, 1-8
12. Successive treatments with ivermectin (3.15%) to control the tick Rhipicephalus (Boophilus) microplus in cattle: Pharmacokinetic and efficacy assessment
Macarena Sarli, Maria Victoria Miro, Maria Victoria Rossner, Santiago Nava, Adrian Lifschitz
Ticks and Tick-borne Diseases, Volume 13, Issue 1, January 2022, 101848
13. Pharmacokinetics of Meropenem in Critically Ill Patients with Acute Renal Failure Treated by Continuous Hemodiafiltration
W. A. Krueger, T. H. Schroeder, M. Hutchison, E. Hoffmann, H.-J. Dieterich, A. Heininger, C. Erley, A. Wehrle, K. Unertl
Antimicrobial Agents and Chemotherapy, Vol. 42, No. 9, Research Article, 17 December 2020
14. A Pharmacokinetic Study of Native E-coli Asparaginase for Acute Lymphoblastic Leukemia Treated with ThaiPOG Protocol
Phumin Chaweephisal, Trai Tharnpanich, Aphinya Suroengrit, Pattramon Aungbamnet, Panya Seksarn, Darintr Sosothikul, Supanun Lauhasurayotin, Kanhatai Chiengthong, Hansamon Poparn, Piti Techavichit
Asian Pac J Cancer Prev. 22 (10), 3309-3315, Oct. 22, 2021
15. Phase I clinical trial repurposing all-trans retinoic acid as a stromal targeting agent for pancreatic cancer
Hemant M. Kocher, Bristi Basu, Fieke E. M. Froeling, Debashis Sarker, Sarah Slater, Dominic Carlin, Nandita M. deSouza, Katja N. De Paepe, Michelle R. Goulart, Christine Hughes, Ahmet Imrali, Rhiannon Roberts, Maria Pawula, Richard Houghton, Cheryl Lawrence, Yathushan Yogeswaran, Kelly Mousa, Carike Coetzee, Peter Sasieni, Aaron Prendergast & David J. Propper
Nature Communications volume 11, Article number: 4841 (2020)
16. SARS-CoV-2 Neutralization in Commercial Lots of Plasma-derived Immunoglobulin
Volk Andreas, Covini-Souris Caroline, Kuehnel Denis, De Mey Christian, Römisch Jürgen, Schmidt Torben
COVID-19 SARS-CoV-2 preprint from bioRxiv, Aug. 13, 2021.
17. Self-emulsifying drug delivery system of (R)-alpha-lipoic acid to improve its stability and oral absorption
Sujan Banik, Shimul Halder, Hideyuki Sato, Satomi Onoue
Biopharmaceutics and Drug Disposition, Vol. 42, Issue 5, May 2021, p. 226-233.
18. The GABA(B) receptor positive allosteric modulator COR659: in vitro metabolism, in vivo pharmacokinetics in rats, synthesis and pharmacological characterization of metabolically protected derivatives.
Francesca Ferlenghi, Paola Maccioni, Claudia Mugnaini, Antonella Brizzi, Federica Fara, Rafaela Mostallino, Maria Paola Castelli, Giancarlo Colombo, Marco Mor, Federica Vacondio, Federico Corelli.
European J. of Pharm. Sci., Vol. 155, 1 Dec. 2020, 105544.
19. Effect of e-cigarette flavors on nicotine delivery and puffing topography: results from a randomized clinical trial of daily smokers.
Voos N, Smith D, Kaiser L, Mahoney MC, Bradizza CM, Kozlowski LT, Benowitz NL, O'Connor RJ, Goniewicz ML.
Psychopharmacology (Berl). 2020 Feb;237(2):491-502. doi: 10.1007/s00213-019-05386-x. Epub 2019 Nov 26.
20. Taylor D.L. et al. (2019) Harnessing Human Microphysiology Systems as Key Experimental Models for Quantitative Systems Pharmacology.
In: Barrett J., Page C., Michel M. (eds) Concepts and Principles of Pharmacology. Handbook of Experimental Pharmacology, Vol 260. Springer, Cham.
21. Long-Acting HIV-1 Fusion Inhibitory Peptides and their Mechanisms of Action.
Chen Wang, Shuihong Cheng, Yuanyuan Zhang, Yibo Ding, Huihui Chong, Hui Xing, Shibo Jiang, Xuebing Li, and Liying Ma.
Viruses, v.11(9); 2019 Sep, PMC6784077
22. Randomized within-subject trial to evaluate smokers initial perceptions, subjective effects and nicotine delivery across six vaporized nicotine products
Natalie Voos, Lisa Kaiser, Martin C. Mahoney, Clara M. Bradizza, Lynn T. Kozlowski, Neal L. Benowitz, Richard J. O'Connor, Maciej L. Goniewicz.
Addiction, 9-Mar-2019, Research Report, Society for the study of Addiction.
23. Safety and Pharmacokinetics of Multiple Doses of Aclidinium Bromide, a Novel Long-Acting Muscarinic Antagonist for the Treatment of Chronic Obstructive Pulmonary Disease, in Healthy Participants
Journal of Clin. Pharm., Vol. 49, Issue 10, 2009, 1239-1246.
Josep M. Jansat PhD, Dr Rosa Lamarca PhD, Dr Gonzalo de Miquel MD, Dr Andreas Schrodter MD, PhD, Dr Barbara Miletzki MD, Mr Matthias Gurniak MSc
24. Aluminium toxicokinetics after intramuscular, subcutaneous, and intravenous injection of Al citrate solution in rats.
Weisser, K., Goen, T., Oduro, J.D. et al,
Arch Toxicol. pp. 1-11 (2018)
25. A Peptide-Based HIV-1 Fusion Inhibitor with Two Tail-Anchors and Palmitic Acid Exhibits Substantially Improved In Vitro and Ex Vivo Anti-HIV-1 Activity and Prolonged In Vivo Half-Life
Shan Su, Giselle Rasquinha, Lanying Du, Qian Wang, Wei Xu, Weihua Li,
Lu Lu and Shibo Jiang
Molecules 2019, 24, 1134 (13 pages).
26. Imperatorin exhibits anticancer activities in human colon cancer cells via the caspase cascade
Yi Mei Zheng, Amy Xiaoxu Lu, James Zheng Shen, Amy Ho Yan Kwok, Wing Shing Ho
Oncology Reports, Jan. 21st 2016, pp 1995-2002.
27. Nicotine Intake from Electronic Cigarettes on Initial Use and After 4 Weeks of Regular Use
Peter Hajek, PhD Maciej L. Goniewicz, PharmD, PhD Anna Phillips, MSc Katie Myers Smith, PhD Oliver West, MSc Hayden McRobbie, PhD
Nicotine Tob Res (2015) 17 (2): 175-179.
28. Fluticasone furoate, a novel inhaled corticosteroid, demonstrates prolonged lung absorption kinetics in man compared with inhaled fluticasone propionate.
Philippe J Bareille; A. Allen; V.M Rousell
GlaxoSmithKline.
MedLine Citation: PMID: 23184737
Clinical pharmacokinetics, Volume: 52,2013 Jan, Clin Pharmacokinet.
29. Anti-oxidant and anti-cancer activities of Angelica dahurica extract via induction of apoptosis in colon cancer cells
YiMeiZhenga, James Zheng Shenb, Yan Wangb, Amy Xiaoxu Lub, Wing Shing Ho
Phytomedicine: international journal of phytotherapy and phytopharmacology 23(11) Dec.2015.
30. The pharmacokinetics of Casodex enantiomers in subjects with impaired liver function
D. Cockshott, E. A. Sotaniemi, K. J. Cooper and D. C. Jones, Zeneca Pharmaceuticals, Alderley Park, Macclesfield, Cheshire and Department of Internal Medicine, University of Oulu, Oulu, Finland
Br J Clin. Pharmacol. 1993; 36: 339-343
31. Lack of Pharmacologic Interaction Between Paroxetine and Alprazolam at Steady State in Healthy Volunteers
Calvo, Gonzalo MD, PhD; Garcia-Gea, Consuelo BSc; Luque, Antonio MD; Morte, Adelaida MD; Dal-Re, Rafael MD, PhD; Barbanoj, Manel MD, PhD
Journal of Clinical Psychopharmacology:
June 2004 - Volume 24 - Issue 3 - pp 268-276
32. A pharmacoscintigraphic study of three time-delayed capsule formulations in healthy male volunteers
McConville JT1, Hodges LA, Jones T, Band JP, O'Mahony B, Lindsay B, Ross AC, Florence, AJ, Stanley AJ, Humphrey MJ, Wilson CG, Stevens HN.
J Pharm Sci. 2009 Nov;98(11):4251-63. doi: 10.1002/jps.21739.
33. Pharmacological characteristics and side effects of a new galenic formulation of propofol without soyabean oil
M. Paul,M. Dueck,S. Kampe,H. Fruendt,S. M. Kasper
First published: 20 October 2003 Full publication history, DOI: 10.1046/j.1365-2044.2003.03345
34. Pharmacokinetic optimization of immunosuppressive therapy in thoracic transplantation:
C. Monchaud, P. Marquet.
Part I. Clin. Pharmaco., Springer Verlag, 2009, 48 (7), pp.419-62.
35. Inflammation-Mediated Abrogation of Androgen Signaling: An In Vitro Model of Prostate Cell Inflammation
Bilge Debelec-Butuner, Cansu Alapinar, Lokman Varisli, Burcu Erbaykent-Tepedelen, Syed Muhammad Hamid, Ceren Gonen-Korkmaz and Kemal Sami Korkmaz
Molecular Carcinogenesis 53:85-97 (2014)
36. Single-Dose, Randomized, Crossover Bioequivalence Study of Amlodipine Maleate versus Amlodipine Besylate in Healthy Volunteers
Fiorenzo Mignini, Daniele Tomassoni, Enea Traini & Francesco Amenta
Pages 539-552 | Received 14 May 2007, Accepted 26 Jun 2007, Published online: 03 Jul 2009
Download citation http://dx.doi.org/10.1080/10641960701744046
37. The GPVI Fc Fusion Protein Revacept Reduces Thrombus Formation and Improves Vascular Dysfunction in Atherosclerosis without Any Impact on Bleeding Times
Martin Ungerer, Zhongmin Li, Christine Baumgartner, Silvia Goebel, Jasmin Vogelmann, Hans-Peter Holthoff, Meinrad Gawaz, Gotz Munch
Published: August 12, 2013
http://dx.doi.org/10.1371/journal.pone.0071193
38. Effects of Recombinant Human Insulin-Like Growth Factor I (IGF-I) Therapy on the Growth Hormone-IGF System of a Patient with a Partial IGF-I Gene Deletion
Cecilia Camacho-Hubner Katie A. Woods Farideh Miraki-Moud Peter C. Hindmarsh Adrian J. Clark Yngve Hansson Atholl Johnston Robert C. Baxter Martin O. Savage
The Journal of Clinical Endocrin. & Metab. Vol. 84, No. 5 1611-1616 (1999) 84 (5): 1611-1616.
39. A Multicenter Study of the Pharmacokinetics of Tacrolimus Ointment after First and Repeated Application to Children with Atopic Dermatitis
John Harper, Catherine Smith, Andris Rubins, Adrian Green, Karina Jackson, Sanita Zigure,
John Bourke, Augusti Alomar, Paul Stevenson, Claire Foster, and Nasrullah Undre
Great Ormond Street Hospital for Children, London, UK;
Skin Therapy Research Unit, University Hospital Lewisham and Kings College London, London, UK; Medical Academy of Latvia, Riga, Latvia; South Infirmary Victoria Hospital, Cork, Ireland;
Hospital de la Santa Creu i Sant Peu, Barcelona, Spain;Fujisawa GmbH, Munich, Germany
J Invest Dermatol 124:695-699, 2005
40. Comparison of the Pharmacokinetic and Pharmacodynamic Profiles of One US-Marketed and Two European-Marketed Epoetin Alfas: A Randomized Prospective Study
Michael Lissy, Marite Ode, and Karsten Roth
Drugs R D. 2011 Mar; 11(1),61-75.
Published online 2012 Nov 27. doi: 10.2165/11588270-000000000-00000
PMCID: PMC3586119
41. Polyinosinic Acid and Polycationic Liposomes Attenuate the Hepatic Clearance of Circulating Plasmid DNA
Rodney F. Minchin, Denise Carpenter and Rebecca J. Orr
Journal of Pharmacology and Experimental Therapeutics March 2001, 296 (3) 1006-1012;
42. Absorption, Distribution, Metabolism and Excretion of 2,3-Dimethyl-2,3-Dinitrobutane.
John. Jenner, Christopher. H. Dalton, Haley. Bird and Stuart. G. Wood.
Biomedical Sciences Department, Dstl, Porton Down, Salisbury. SP4 0JQ, UK
Biodynamics Ltd, Pegasus Way, Rushden, Northants. NN10 6ER, UK, (C) Crown 2004
43. The pharmacokinetics, metabolism and urinary detection time of tramadol in camels
M. Elghazali, I.M. Barezaik, A.A. Abdel Hadi, F.M. Eltayeb, J. Al Masri, I.A. Wase
The Veterinary Journal 178 (2008) 272-27
44. Pharmacokinetic, metabolism and withdrawal time of orphenadrine in camels (Camelus dromedarius) after intravenous administration
M. Elghazali
The Veterinary Journal Oct (2016)
45. Absorption, metabolism and excretion studies on clavulanic acid in the rat and dog.
Allen G.D., Bolton G.C., Filer C.W. and Jeffery D.J.
Xenobiotica, Vol. 14, No. 6, 483-490, 1984.
46. The disposition of clavulanic acid in man.
Allen G.D., Bolton G.C., Davies B.E., Filer C.W. and Jeffery D.J.
Xenobiotica, Vol. 16, No. 9, 853-863, 1986.
47. On the absorption of clavulanic acid.
Allen G.D., Coates P.E. and Davies B.E.
Biopharmaceutics and Drug Disposition, Vol. 9, 127-136, 1988.
48. Absorption, metabolism and excretion of C14 denbufylline in man after i.v. and oral administration.
Allen G.D., Koester F.E., Langley P.F. and Palm M.
British Journal of Clinical Pharmacology, 33, 236, 1992.
49. Investigation of the single and repeat dose oral pharmacokinetics of denbufylline in healthy young and elderly subjects.
Allen G.D., Brett M.A., Gulati R., Hoetzel L., Hollis F.J., Pfeiffer P. and Thawley A.R.
British Journal of Clinical Pharmacology, 33, 235, 1992
50. Metabolic and PK studies following oral administration of C14 famciclovir to healthy subjects.
Allen G.D., Brown T.A., Filer C.W., Fowles S.E., Mort E., Prince W.T. and Ramji J.V.
Xenobiotica, 24(4), 357-368, 1994.
51. Disposition of famciclovir in healthy volunteers.
Filer C.W., Allen G.D., Mort E., Fowles S.E. and Ramji J.V.
6th International congress for infectious diseases, Prague, March, 1994.
52. Quantitative HPLC-MS-MS in support of a pharmacokinetic study involving simultaneous constant rate and accelerated infusions via the intravenous and intraportal routes.
Allen G.D., Griffiths R., Abbott R.W., Bartlett S., Brown T., Lewis V.A., Nash M. and Rontree J.
GlaxoSmithKline.
Proceedings of 13th International MS Conference, Budapest, September, 1994.
International Biomedical Symposium, Life Sciences, California, Sept. 1994
53. Phase I double blind, placebo controlled, ascending single intravenous dose study with a synthetic direct thrombin inhibitor, TGN255.
Combe S, Allen G.D. and Kennedy T.
The 18th International Congress on Thrombosis (ICT), June 20-24, 2004, Ljubljana, Slovenia.
54. A Phase I double blind 24-hour i.v. infusion study with TGN255, a direct thrombin inhibitor.
Combe S, Allen G.D. and Kennedy T., ASN (27 Sept.-1 Oct. 2004) Washington DC, USA.
55. Pharmacokinetics of TGN 255, a Novel Intravenous Low Molecular Weight Direct Thrombin Inhibitor, In Healthy Volunteers
Allen G.D., Combe S, Dupe R.and Kennedy T.
Poster P1714 at the XXth Congress of the International Society of Thrombosis and Haemostasis (ISTH), 6-12 August 2005, Sydney, Australia.
56. Comparative Pharmacodynamics of TGN 255, a Novel Direct Thrombin Inhibitor and Unfractionated Heparin in Healthy Volunteers
Combe S, Dupe R, Allen G. and Kennedy T.
Poster 1864 at the 47th Annual Meeting of the American Society of Hematology, 2-6 December 2005, Atlanta, Georgia, USA.
57. A Phase I Double-Blind, Ascending Dose Study of an Oral Synthetic Direct Thrombin Inhibitor, TGN167.
Combe S, Kennedy T, Allen G
Blood (ASH Annual Meeting Abstracts) 2005 106: Abstract 1863
58. A Phase I Interaction Study between TGN 255, a Direct Thrombin Inhibitor, and Aspirin plus Clopidogrel
Combe S, Kennedy T, Allen G
Poster Presentation at the World Congress of Cardiology, 2-6 September 2006, Barcelona, Spain.
59. Pharmacokinetics of F901318 in man from an Intravenous Single Ascending Dose Study.
T. Kennedy, J. Steiner, G.D. Allen, J. Oliver, M. Birch, G. Sibley, D. Law
Interscience Conf. Antimicrob. Agents and Chem. ICAAC 17-21 Sept. 2015. San Diego, California.
60. The relationship between paroxetine and the sparteine oxidation polymorphism.
Sindrup SH, Brosen K, Gram LF, Hallas J, Skjelbo E, Allen A, Allen GD, Cooper SM, Mellows G, Tasker TC, et al.
Clin Pharmacol Ther. 1992 Mar;51(3):278-87.
61. A Multicenter Study of the Pharmacokinetics of Tacrolimus Ointment after First and Repeated Application to Children with Atopic Dermatitis
Undre N et al
Journal of Investigative Dermatology (2005) 124, 695-699
62. Influence of albumin supplementation on tacrolimus and cyclosporine therapy early after liver transplantation
Andrew Trull, PhD, Victoria Hughes, Dawn Cooper, Matthew Wilkins, Alexander Gimson, Peter Friend, Atholl Johnston, Linda Sharples, Gilbert Park
Liver Transplantation
Volume 8, Issue 3, Pages 224-232, Published Online: 30 Dec 2003
Copyright (C) 2002 by the American Association for the Study of Liver Diseases
63. Pharmacokinetics of S- and R-Enantiomers of Aminoglutethimide following Oral Administration of Racemic Drug in Breast Cancer Patients
Dr. Ibrahim A. Alshowaier PhD, Dr. Adnan El-Yazigi PhD, FCP, Dr. Adnan Ezzat MD, Dr. Ahmad Abd El-Warith MD, Dr. IP. J. Nicholls PhD
Pharmacokinetics Laboratory, Department of Biological and Medical Research, MBC-03, King Faisal Specialist Hospital and Research Centre, Riyadh 11211, Saudi Arabia
J. Clin. Pharmacol. November 1999
64. Patent: Prodrugs of Substituted 1,3-Dioxanes and Their Uses, US 20150158838 A1
65. Patent: Thrombin inhibitor, CA 2691243 A1
66. Prediction of Plerixafor Pharmacokinetics in Mice Using PCModfit Software
Sihana AHMETI LIKA Dorentina BEXHETI, Edita Alili IDRIZI
2018, 3rd International Scientific Conference of the Medical Sciences - University of Tetova
67. Pharmacokinetic optimization of immunosuppressive therapy in thoracic transplantation: part I
Caroline Monchaud and Pierre Marquet
Clin Pharmacokinet. 2009; 48(7): 419-462.
68. IgG Fc-binding motif-conjugated HIV-1 fusion inhibitor exhibits improved potency and in vivo half-life: Potential application in combination with broad neutralizing antibodies
Wenwen Bi, Wei Xu, Liang Cheng, Jing Xue, Qian Wang, Fei Yu, Shuai Xia, Qi Wang, Guangming Li, Chuan Qin, Lu Lu , Lishan Su , Shibo Jiang
2019 (5 Dec.) Research Article, PLOS Pathogens 15 (12) e1008082.https://doi.org/ 10.1371/journal.ppat.1008082